|
Clearly, the characterization of cathode rays was a process begun long before Thomson's work, and several scientists made important contributions. The case of the electron raises several interesting points about the discovery process. He was awarded the Nobel Prize for physics in 1906 for this work, and in 1908 he was knighted. In 1899, he measured the charge of the particles, and speculated on how they were assembled into atoms. In 1897 he reported that "cathode rays" were actually negatively charged particles in motion he argued that the charged particles weighed much less than the lightest atom and were in fact constituents of atoms. For much of his career, Thomson worked on various aspects of the conduction of electricity through gases. Thomson was the Cavendish professor of Experimental Physics at Cambridge University and director of its Cavendish Laboratory from 1884 until 1919. Thomson, 1856-1940 see photo at American Institute of Physics) is widely recognized as the discoverer of the electron. Thomson found that this value comes to \( 1.76 \times 10^8 C/g \).Discovery of the Electron: J. The charge-to-mass ratio of the electron can be determined by measuring the effects of the magnetic and electric fields on the motion of the beam.What is the charge-to-mass ratio for the cathode ray particles?.If the correct combination is reached, the can completely cancel each other out and not deflect the rays at all. When both electric and magnetic forces are applied to the cathode ray, they combine against or with each other to deflect the cathode rays. 
What is the effect of simultaneous electric and magnetic fields on the cathode ray?. 
Again, this is due to the negative charge the cathode rays possess. When the north pole is facing towards the CTR, the beam is deflected upwards. If the south pole is facing towards the CTR (Cathode Ray Tube), the beam is deflected down. When a magnetic field is applied, the cathode rays get deflected.What is the effect of a magnetic field on the cathode ray?.This is because the cathode rays are negatively charged. When an electric field is applied to a stream of cathode rays, they are deflected towards the positive plate.What is the effect of an electric field on the cathode ray?.Notice the dotted circle indicating the position of the coils, the direction of the magnetic field between the coils and the value of the field. In the Thomson applet, set the voltage at 0 V and move the slider for the current to the right. When a current flows through the coils, a uniform magnetic field is created in the region between the horizontal plates. Using the same tube as shown in Figure 1 (above), he placed a pair of coils outside of the tube and on either side of the horizontal plates (Figure 3, below). 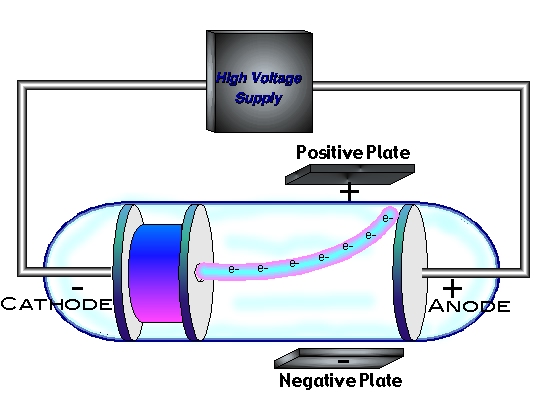 
Thomson also used a magnetic field to affect the beam of cathode rays. In the Thomson applet, this angle is provided by selecting Deflection Angle in the Options menu. From measurements of this deflection, he was able to calculate the angle of deflection. Thomson measured the deflection of the beam using a ruler etched on the end of the tube. You can see a simulation of this glow on the far right of the applet diagram, as shown in Figure 2. In Thomson’s experiment, a fluorescent material was coated on the end of the tube to produce a glowing dot where the cathode rays hit.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
